Improved method for inducing chronic atrophic gastritis in mice
2019-12-14XianWeiXuePingFengLuYaoWangYanQiangHuangLingLingLiangXiaoQiangMoHongYuWei
Xian Wei,Xue-Ping Feng,Lu-Yao Wang,Yan-Qiang Huang,Ling-Ling Liang,Xiao-Qiang Mo,Hong-Yu Wei
Abstract
Key words: Method; Chronic atrophic gastritis; Mice; Helicobacter pylori; N-methyl-N'-nitroguanidine; Ammonia water
INTRODUCTION
Chronic atrophic gastritis (CAG) is a common disease of the digestive system with pathological characteristics of a decreasing number,or disappearance,of inherent glands of gastric mucosa[1,2].As early as 1978,the World Health Organization had announced a close relationship between CAG and the incidence of gastric cancer[3].CAG has been defined as a precancerous condition of gastric cancer.Intestinal metaplasia or intraepithelial neoplasia accompanying atrophied glands of the stomach is regarded as one of the most important precancerous lesions of gastric cancer.As a common malignant tumour,gastric cancer remains without a satisfactory therapy and its pathogenesis remains unclear,seriously threatening human life[4,5].Therefore,some scholars have proposed to prevent the incidence of gastric cancer by avoiding precancerous lesions.If CAG can be reversed,the incidence of gastric cancer can be substantially reduced.To reverse and prevent CAG and study its pathogenesis and therapy,it is necessary to develop an ideal,safe,stable,animal model.An ideal animal model of CAG needs to meet the following conditions: (1) The model develops disease to resemble the same condition in humans; (2) Animals are abundant and easily raised; and (3) The model is simple and provides a high success rate,and readily guarantees survival of the animals.In recent years,scholars mainly established animal models by imitating human CAG etiology.Commonly used animals include rats and Mongolian gerbils.Methods based on liveHelicobacter pylori(H.pylori),N-methyl-N'-nitroguanidine (MNNG),ammonia,sodium deoxycholate,sodium salicylate,and immunology are commonly used to establish animal models[6-8].In addition to this,the animal model can also be developed through combination of diseases and syndromes according to traditional Chinese medicine(TCM)[9].Regardless of what method is used,the results differ to some extent from the ideal animal model and certain deficiencies therein require to be overcome.It is a challenge to build a CAG model with mice.For example,even if the methods of chemical mutagens and stimulation of the gastric mucosa with a combination of factors are used for establishing animal models with a high success rate,they retain certain shortcomings.These include various problems,such as the selection of experimental animals,drug combination,drug dosages,and lack of a unified standard for the duration of the modelling.Besides,spontaneous animal models are seldom studied.TCM diagnoses and treats CAG mainly based on an overall analysis of patient condition,rather than the method applied by Western medicine that treats CAG in a general way according to the classification of diseases.Therefore,it is a preferable research approach to establish experimental animals combining diseases and syndromes using TCM to carry out TCM basic and clinical research.Even so,there are also some drawbacks.For instance,there is a lack of a recognised method of developing models and objective evaluation indices thereof.Therefore,it is necessary to improve the animal model of CAG,and explore an ideal method of developing mouse models of CAG.H.pyloriis the primary pathogenic factor of CAG; the MNNG method is an acknowledged method of making mouse models of CAG; and ammonia simulates the injury of alkaline environment to the gastric mucosa,so it is also a clinically common method for establishing animal models based on etiology.Considering this,this study proposed an improved comprehensive method for establishing an animal model by combiningH.pylori,MNNG,and ammonia.To our knowledge,the combined modelling method has not yet been reported.
MATERIALS AND METHODS
Animals
Female six-week-old Kunming mice,weighing between 180 and 200g,were purchased from the Experimental Animal Center,Youjiang Medical University for Nationalities.The animals were housed at controlled humidity (50% ± 5%) and temperature (25±2°C) with a light-dark cycle of 12h.Animal treatment was in accordance with National Institute of Health guidelines and experimental protocols were approved by the Institutional Animal Care and Use Committee of Youjiang Medical University for Nationalities.
Materials
The study involvedH.pyloristrains (Affiliated Hospital of Youjiang Medical University for Nationalities),MNNG (BIDE PHARMATECH Co.,ltd),ammonia water(Sinopharm Chemical Reagent Co.,Ltd.),IL-1β,IL-8,TNF-α,CD4,CD8,CagA,Bax,a Bcl-2 kit (Beijing Solarbio Science and Technology Co.,ltd.),and a DNA kit(TIANGEN,BIOTECH Co.,ltd.).All reagents were of analytical grade.
Animal groups
A total of 220 six-week-old Kunming mice were randomly divided into a phosphate buffered solution control group,anH.pylorigroup,an MNNG group,an ammonia water group,and a group combiningH.pylori,MNNG,and ammonia water(hereinafter referred to as the combined group).Each group contained equal numbers of male and female mice.
Mutagenesis and injury by MNNG
Mice in the MNNG and combined groups were administrated with MNNG (5 mL/kg body mass) at a concentration of 120 μg/mL through daily gavage for seven successive days.
Inducing injury and neutralising gastric acid by ammonia water
Mice were fed drinking water containing 0.02 wt% ammonia throughout.Mice in the control group were fed regularly without intervention.
H.pylori infection
Clinically isolatedH.pyloristrains were used,and they were subjected to Gram staining,urease testing,oxidase testing,and catalase testing,and identified asH.pylorithrough 16S ribosomal RNA sequencing.Strains were CagA and VagA positive and were proliferated and cultured for three days.The concentration of biosynthetic human insulin (BHI) medium was adjusted to 1 × 109CFU/mL.H.pyloriwas applied to the mice through gavage after intragastric administration of MNNG.The mice were fasted for 12 h before gavage.The mice in theH.pyloriand combined groups were intragastrically administrated with 5 × 108CFU ofH.pylori.Thereafter,the mice were fasted for solids and liquids for 2 h.The administration was conducted every other day,five times in succession.In comparison,0.5 mL of BHI medium was applied to each mouse in the control group through gavage (also five times).
Confirming modelling results
Five male and five female mice were chosen from each group on the 30th,60th,90th,and 120th days.Then,gastric mucosal samples were collected for pathological examination by hematoxylin eosin (HE) staining,terminal nick-end labelling(TUNEL) staining,and immunohistochemical staining for Bax and Bcl-2.In addition,H.pyloriwas isolated,cultured,and identified,and the extent of its colonisation calculated.Blood was collected to detect the inflammatory factors IL-1β,IL-8,and TNF-α,and immune function markers CD4 and CD8.Besides,the liver,kidney,and spleen samples of the modelling groups were detected by HE staining to confirm whether or not CAG has been successfully induced.
RESULTS
HE-stained gastric mucosa
On the 30th day after infection,all of the mice in the combined group were found to have developed acute gastritis,while there were no CAG mice.No significant difference was shown on the 60th day compared with that on the 30th day.On the 90th day,90% (nine out of ten) of mice had developed slight CAG which developed to moderate CAG on the 120th day,as shown in Figure 1.
TUNEL-stained gastric mucosa
The detection of TUNEL-stained gastric mucosa of mice in the combined group on the 30th,60th,90th,and 120th days after infection revealed that epithelial cells of the gastric mucosa all presented significant apoptosis.There was greater cell apoptosis by the 30th day,while at the 90th and 120th days,similar,less severe,apoptosis than that at the 30th day was found,as shown in Figure 2.
Immunohistochemical staining for Bax and Bcl-2
Bax,directly activated by p53,can improve apoptotic sensitivity and promote p53-mediated cell apoptosis.Bcl-2 protein can form a heterodimer with other proapoptotic proteins to inhibit cell apoptosis.It also plays an anti-apoptosis effect by interacting with apoptotic protease activating factor or inhibiting the release of cytochrome c,an activator of mitochondrial caspases.Therefore,cell apoptosis can be examined by detecting the expression of Bcl-xL and Bax.The expression of Bax in the combined group was up-regulated at the 90th and 120th day,while that of Bcl-2 was down-regulated,which indicated that the mice in the combined group had undergone significant cell apoptosis,conforming to the characteristics of CAG,as shown in Figure 3.
Identification and colonisation of isolated cultures
According to the postulates of Robert Koch,infectingH.pylorihas to be isolated and cultured from the modelling groups,so the gastric mucosa was collected for isolation and culture ofH.pylori,and the cultures were identified.The specific results are described below.
Gram staining:Isolated cultures were selected for Gram staining.The bacteria were Gram negative under a microscope and curving,which is consistent with the morphological characteristics of stainedH.pylori,as shown in Figure 4.
CagA detection:CagA is a typical virulence factor ofH.pylori.It had been verified that the infectingH.pyloristrains contain the CagA gene beforeH.pyloriinfection through gavage.Therefore,if the isolated bacterium wasH.pylori,the genome was bound to contain the CagA gene.It was verified that the isolated cultures did contain the CagA gene,as shown in Figure 5.
Detection of oxidase,peroxidase,and urease:H.pylorishows the biochemical characteristic of positive oxidase,peroxidase,and urease,so the three factors are commonly used as biochemical indices for identifyingH.pylori.Detection of the isolated cultures showed that the three were also positive,which accorded with the biochemical characteristics ofH.pylori,as summarised in Table 1.
Extent of H.pylori colonisation:The above identifications revealed that the isolated and cultured bacterium wasH.pylori,whose population was calculated then to estimate the extent of its colonisation.At the 30th,60th,90th,and 120th days after infection,every gram of gastric mucosa of mice in the combined group was found to contain 1 × 104to 1 × 105CFU ofH.pylori,and there was no significant difference across different time periods.This indicated that the colonisation ofH.pyloridid not decrease within 120 d afterH.pyloriinfection,soH.pyloriprobably played a significant role in the pathogenic process of CAG,as shown in Figure 6.

Figure 1 HE-stained gastric mucosal tissues of mice in the combined group (magnification,×100).
Detection of inflammatory factors
The incidence and development of CAG can trigger the release of various inflammatory factors such as IL-1β,IL-8,and TNF-α.Inflammatory factors IL-1α,IL-8,and TNF-α were found to have increased expression on the 30th,90th,and 120th days after mice in the combined group were infected withH.pylori.The increase was greater the first month,while the inflammatory factors had equivalent expression at the 90th and 120th days,both being lower than that on the 30th day.The results are consistent with the results of TUNEL staining,also showing the up-regulation of CAG inflammatory factors,as shown in Figure 7.
Detection of immunological functions
The expression of CD4+ and CD8+ immune cells of mice in the combined group were both decreased by the 30th,90th,and 120th days after infection.The expression was down-regulated significantly by the 30th day,while the two immune cells were expressed in a similar manner on the 90th and 120th days,both having been downregulated to a lesser extent than that on the 30th day.This was mainly because,by the 30th day,the mice were still affected by the immunosuppressor MNNG,and their immunological functions were impaired.After eliminating the influences of the immunosuppressor,the immunological functions improved while were unable to recover to normal levels.This is also probably because CAG itself is characterised by the impairment of immunological function,as seen from Table 2.
Impairment of other organs
As chemical and biological methods were combined in the study to establish the model,chemical reagents were likely to lead to impairment of organ functions of the mice,and even cancers therein.Therefore,it is necessary to detect the impairment of organs of the mice.By collecting and staining the liver,kidney,and spleen tissues with HE,it revealed that there was no impairment.This indicated that the modelling method did not induce complications of the liver,kidney,and spleen except for the development of CAG,so the method could be deemed safe,as shown in Figure 8.
心理情境创设:心理情境是唤起学员身体感觉、激发积极情绪的重要途径。所以在培训中提倡“走动式教学”,避免教师一成不变地坐在讲台上授课。研究表明,我们的精神无法和身体分开,物理距离会激发心理疏远的感觉(Blass 1991、Mobbs 2007),所以只有走入到学员中,才能真正拉近师生之间的距离,建立良好的教学情感,并能及时关注学员的教学所需。另外,走动也帮助教师运用更多的肢体语言、眼神与学员们进行互动,激发学员们良好的情绪体验。
Statistics of successful mice models
The aforementioned identifications proved that the established animal models were CAG models.Statistical analysis revealed that 90% of mice in the combined group developed CAG at the 90th and 120th day after infection andH.pyloricolonisation was found in all such mice.In contrast,theH.pylorigroup showedH.pyloricolonisation but no CAG.Other groups did not exhibit CAG orH.pyloricolonisation,as can be seen from the data in Table 3.
DISCUSSION
CAG is one of the most common gastrointestinal diseases and an early stage of gastric cancer.CAG is actually one event in a chronic degenerative process beginning with gastritis,proceeding to CAG,then to intestinal metaplasia,dysplasia and,finally,to carcinoma[10].CAG is hard to be cured with recurrent attacks,severely affecting the health of human.The research and treatment of CAG are very popular.An animal model of CAG is an important basis for research into chronic gastritis.Although there have been numerous reports on the animal models of CAG,it is still not clear which model is best suited to investigating the mechanism of occurrence of human chronic gastritis and there are no unified standards for the type and age of animals to be selected[11-13].As the modelling of chronic gastritis takes a long time,there is an urgent need to find a more rapid,stable,safe model.At present,biological modelling,chemical injury,and active immunisation are commonly used methods of establishing animal models,and rats are generally used.MNNG,sodium deoxycholate,and sodium salicylate are commonly model materials with chemical methods.Rats and Mongolian gerbils are commonly used animals to develop CAG models,while mice are unusually used.The reasons may be that mice do not control the dose of the intervention and are prone to other complications or toxic side effects.
H.pyloriis a human-specific pathogen,which leads to gastric pathologies including gastritis and gastric ulcers.H.pyloriis a common risk factor for CAG and can be used to create CAG model.The animal model is due toH.pyloriinfection,more like a human infection model,and beneficial to studying the pathogenesis of CAG.However,H.pyloriis relatively easy to implant in rats and Mongolian gerbils,and the incidence rate is relatively high,but it is difficult to plant in the mouse.Clinically isolatedH.pyloristrains must be domesticated for adapted planting,and the time of retaining in the stomach is relatively short.After the first month of infection,the maximum number of plants is reached,and it will decrease in the second and third months.The mice will not be able to maintain a relatively high infection rate.Although the C57BL/6 mice and BALB/c mice withH.pyloriSydney strain (SS1) can maintain a high amount of plants in the stomach for a long time,the strain was CagA negative and CAG did not appear in 24 wk after the last infection[14].“H.pylori+ Nmethyl-N-nitrourea can induce CAG in 23.1% of animals after infection for 36 wk,which required a long time,and the success rate is low[15].As it is difficult to coloniseH.pyloriin the stomach in the long term,and particularly difficult to infect mice for a long time,biological modelling commonly uses rats.However,H.pyloridoes not readily infect rats without injury to the gastric mucosa,therefore,the gastric mucosa of rats has to be impaired before being infected withH.pyloriin many experiments.So,it is very difficult to establish CAG withH.pyloriinfected mice in a short period of time.
In reference to the methods of Berginet al[16],Nagaharaet al[17],and Jinet al[18],this study established a CAG model with Kunming mice by combiningH.pylori,MNNG,and ammonia water.The method was proposed on the basis of the fact that infection withH.pylorican induce gastritis[19,20],the functions of MNNG as a chemical mutagen and carcinogen[21,22],and the role of ammonia water in neutralising gastric acid and impairing gastric mucosa[23,24].A CAG model was successfully established by the 90th day after the lastH.pyloriinfection of six-week-old mice,with a success rate reaching 90%.The method overcomes the difficulty in colonisingH.pyloriin mice and the fact that the colonisation time is short.In addition,rats are replaced with mice,and the method is thus simpler and also cheaper.The established models are stable and safe,therefore,the method is a relatively ideal method for establishing a mouse model of CAG.It is demonstrated to be possible to build an animal model that is analogous to human CAG,which provides a significant help when exploring the mechanisms of occurrence and prevention of CAG.

Figure 2 TUNEL-stained gastric mucosal tissues of mice in the combined group (magnification,×100).

Table 1 Identification of cultures isolated from gastric mucosa of mice in the combined group based on biochemical indices

Table 2 Immunological functionsof mice in the combined group

Table 3 Modelling results of chronic atrophic gastritis
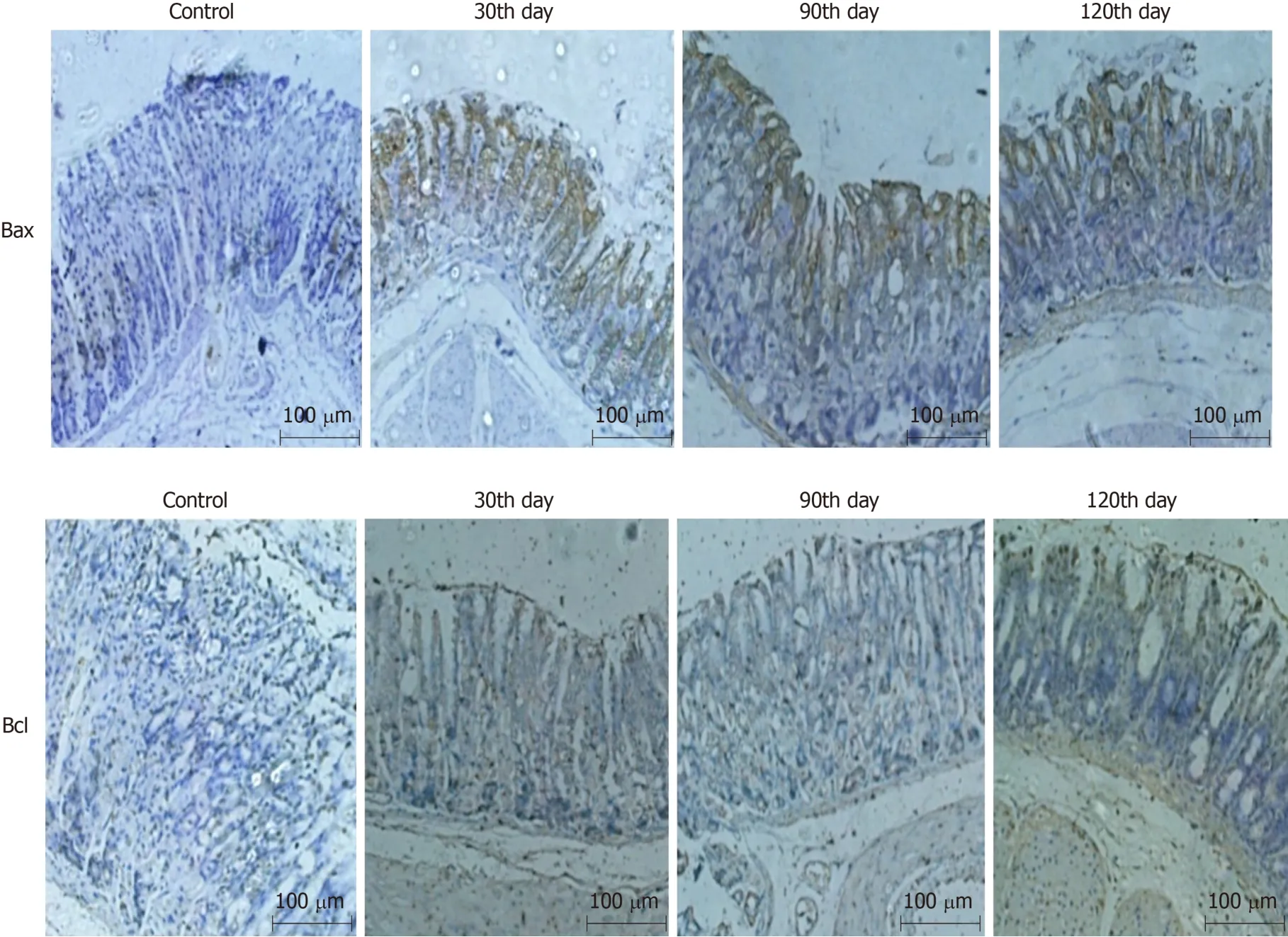
Figure 3 Immunohistochemical staining for Bax and Bcl-2 in the gastric mucosa of mice in the combined group (magnification,×100).

Figure 4 Gram staining of Helicobacter pylori cultures isolated from the gastric mucosa of mice in the combined group (magnification,×100).

Figure 5 Cag A detection for Helicobacter pylori isolated and cultured from the gastric mucosa of mice in the combined group.

Figure 6 Helicobacter pylori colonisation in the gastric mucosa of mice in the combined group.

Figure 7 Inflammatory factors in mice in the combined group.

Figure 8 HE-stained liver,kidney,and spleen tissues of mice in the combined group (magnification,×100).
ARTICLE HIGHLIGHTS
Research background
Chronic atrophic gastritis (CAG) is a precancerous lesion of gastric cancer.If it is effectively treated,GC will be prevented.So the prevention and treatment of CAG are very important.It needs a good animal model,which can provide etiological research and screening of therapeutic drugs.It is an important tool for stu dying CAG.
Research motivation
Current animal models of chronic atrophic gastritis are not ideal,long cycle,high cost,difficult to operate,and unstable,so better animal models are needed.
Research objectives
To construct a more rapid,safe,stable and efficient chronic atrophic gastritis model with mice.
Research methods
The mice selected for this method were six-week-old Kunming mice,and Helicobacter pylori,Nmethyl-N'-nitroguanidine,and ammonia water were combined to develop a CAG model.
Research results
The method showed slight CAG at the 90th day and moderate CAG at the 120th day.
Research conclusions
The method presented here is more rapid,safe,stable,and efficient.
Research perspectives
Starting from the etiology of chronic atrophic gastritis,the course of the disease is simulated to explore a simple,fast,convenient,and stable method of modelling.
猜你喜欢
杂志排行
World Journal of Gastrointestinal Oncology的其它文章
- Observation or resection of pancreatic intraductal papillary mucinous neoplasm: An ongoing tug of war
- Relationship between cachexia and perineural invasion in pancreatic adenocarcinoma
- Protein expression trends of DNMT1 in gastrointestinal diseases: From benign to precancerous lesions to cancer
- Asian Americans have better outcomes of non-metastatic gastric cancer compared to other United States racial groups: A secondary analysis from a randomized study
- Gastric partitioning for the treatment of malignant gastric outlet obstruction
- Difference in failure patterns of pT3-4N0-3M0 esophageal cancer treated by surgery vs surgery plus radiotherapy
