An innovative method used for the identif ication of N-glycans on soybean allergen β-conglycinins
2023-01-23ChengLiYngTinJinliHnYuLuMeiyiZouYueJiChengjinWngLinjunHungZhongfuWng
Cheng Li, Yng Tin, Jinli Hn, Yu Lu, Meiyi Zou, Yue Ji,Chengjin Wng, Linjun Hung,,*, Zhongfu Wng,,*
a Key Laboratory of Glycobiology and Glycoengineering of Xi’an, College of Food Science and Technology, Northwest University, Xi’an 710069, China
b College of Life Sciences, Northwest University, Xi’an 710069, China
Keywords:Soybean allergen β-conglycinin Glycopeptide Mass spectrometry N-Glycan Glycosylation site
A B S T R A C T β-Conglycinin is one of the major allergens existed in soybean. N-Glycans attached to the β-conglycinin influenced the immunoreactivity and antigen presenting efficiency of β-conglycinin. In this study, we described a new method used to release and collect the N-glycans from β-conglycinin, and the N-glycans existed in linear epitopes of β-conglycinin were identifi ed. Glycopeptides hydrolyzed from β-conglycinin were purifi ed by cotton hydrophilic chromatography. Trifl uoromethylsulfonic acid was then used to release glycans from glycopeptides, and new glycopeptides containing one single N-acetyl-D-glucosamine (GlcNAc) moiety were then utilized for mass spectrometry. Five glycosylation sites (Asn-199, Asn-455, Asn-215, Asn-489 and Asn-326) and 22 kinds of glycopeptides were identifi ed. It is noteworthy that the peptide VVN#ATSNL( where # represents for the glycosylation site) was analyzed to be both glycopeptide and linear epitope. Our results provided a new method for the N-glycoform analysis of food allergens, and laid a foundation for understanding the relationship between glycosylation and food allergy.
1. Introduction
As a common and complex post-translational modification,glycosylation altered the protein structures, and then influenced the biological functions of protein molecules [1,2]. Glycoproteins are closely associated with cell proliferation, cell regulation, signal transduction, adhesion, fusion, and pathogen infection [3]. It is worth noticing that many food allergens are glycoproteins. Both glycosylation sites and their corresponding glycans influenced the immunoreactivity of food allergens, which are closely related with the occurrence and development of food allergy [4]. Glycans which recognized by antibodies are named as carbohydrate cross-reactive determinants (CCDs), someN-glycans in plant extracts have been reported to be IgE-binding epitopes [5], and the core region ofN-glycans in honeybee venom allergens were proved to be one of the major allergic epitopes [6].
Besides, glycans attached to the glycoproteins could help the antigen presenting cells (APCs) to recognize the allergens and then transform the immune signals to the following immunologic effector cells [7]. It has been reported that the immunoreactivity of peanut allergen Ara h 1 decreased signif icantly when all the glycans attached to protein were removed, the reason may lay on the fact that dendritic cells had a signif icantly lower uptake of the deglycosylated Ara h 1 [8].Another research had proved that compared with natural ovalbumin,the degranulation rate of basophilic granulocytes decreased significantly when the cells were treated with deglycosylated ovalbumin [9]. For now, researches on food allergens are mainly focused on the structural characteristics of allergic proteins, the immune effect of glycans which attached to the allergens has not been thoroughly studied. Since glycosylation is closely associated with the pathogenesis of food allergy, we think that it is crucial to define theN-glycan structural features and glycosylation sites on allergens.
Unlike protein and DNA biosynthesis, there is no single template for glycan synthesis. Protein glycosylation is regulated by different factors, and the structure of glycans may vary on the same glycosylation site. The microheterogeneity between glycosylation sites and glycan structures make it difficult to analyze the structure ofN-glycans [10]. Enrichment of glycopeptides is the first and essential step in the glycosylation analysis, for the content of glycopeptides accounts for only 2%-5% among all the hydrolyzed peptides [11].At present, glycopeptides are usually enriched by lectin affinity chromatography and chemical methods [12]. Lectins could specifically recognize glycans, and bind them with noncovalent bonding. It cannot be neglected that lectin affinity chromatography is costly, and in some occasions, glycopeptides with complex structures may not be collected efficiently by lectin affinity chromatography. Chemical methods used for glycopeptide enrichment includes hydrazine and boric acid treatments. After attached to the stationary phase, hydrazine was used to oxidize the dihydroxyls on the glycans to form aldehydes, which would further react with the hydrazine to form hydrazones and then bound to the stationary phase carrier [13,14]. However, this method would destroy the natural structure ofN-glycans on the glycopeptides,leading to a missing of information about the glycan structure. Boric acid, which attached to the stationary phase carrier, could prompt the hydroxylation betweencis-adjacent ormeso-adjacent hydroxyls on the glycans and form cyclic diesters [15], cyclic diesters bound to the stationary phase could help to enrich glycopeptides. Butcishydroxynucleotides existed in the reaction system would attach to the solid-phase carrier and distort the experimental results.Glycopeptides could retain on the chromatographic columns by hydrophilic interactions, and impurities with low-hydrophilicity were eluted during the enrichment. Various types of hydrophilic interaction chromatography (HILIC) such as microcrystalline cellulose columns and cellulose microspheres were used to purify glycopeptides and glycans [16]. Given the disadvantages of present methods used for the purification and analysis of glycopeptides, our research group has developed a new cotton-based glycan enrichment strategy [17].Hydroxyl groups on cotton would prompt the combination between cotton and glycopeptides. Compared with other HILIC, cotton HILIC is simple, efficient, easy to prepare, and cost-effective.
As one of the typical food allergens among the world, soybean could cause allergic symptoms like nausea, vomitus, lip swelling, mouth erythema, and even life danger [18]. The prevalence of soybean allergy is estimated to be 0.3% in adults, and 0.1%-0.6% in infants [19,20].Soybean allergenβ-conglycinin, also known as Gly m 5, has been proved to be a typical glycosylated protein, all the glycans were attached to the asparaginase inβ-conglycinin [21]. Glycans attached to the free amino groups of asparaginate are named asN-glycans,while glycans attached to the hydroxyl groups in serine, threonine,and tyrosine are named asO-glycans. In this study, we analyzedN-glycosylation sites and the correspondingN-glycan structure ofβ-conglycinin based on cotton hydrophilic chromatography. New methods used for glycopeptides enriching and glycans release in this research could be recommended for the glycosylation analysis of food allergens. Results from this research could help us to modify the allergen’s glycans in the amino acid level. Detailed structural information about allergens would be helpful for the understanding of molecular mechanism of food allergy, and be further used to develop targeted vaccines and/or therapeutic agents for the prevention and cure of food allergy.
2. Materials and methods
2.1 Reagents and instruments
Peptide N Glycosidase F (PNGase F), trypsin (batch No.slbm2321v), and pepsin (batch No. 091m7020v) were purchased from Sigma Aldrich Co. (St. Louis, MO, USA). Medical-grade absorbent cotton was acquired from Northwest University Hospital (Shaanxi Province, China). Sep-Pak C18solid phase extraction columns(200 mg/4 mL) and SPE columns (150 mg/4 mL) were purchased from Waters Co. (Milford, MA, USA). Trifluoromethylsulfonic acid (TFMS) was purchased from Shanghai Aladdin Reagent Co.(Shanghai, China). The experimental water was prepared in a Milli-Q system (Millipore, Burlington, MA, USA). Bicinchoninic acid (BCA)protein assay kit (batch No. ab102536) was purchased from Beyotime(Shanghai, China). Soybean (Glycine maxL.) were purchased from the supermarket named Darunfa, which located in Laodong South Road, Lianhu district, Xi’an, Shaanxi, China.
2.2 Experimental strategy
Trypsin and pepsin were used to hydrolyze the protein and a mixture of glycopeptide and non-glycopeptide was obtained.Glycopeptides was subsequently collected by cotton hydrophilic chromatography. And then, a part of glycopeptides was treated with PNGase F to release theN-glycans, and other part of the glycopeptides was treated with TFMS to produce glycopeptides containing one singleN-acetyl-D-glucosamine (GlcNAc) moiety (molecular weight 202 Da).By using electrospray ionization mass spectrometry (ESI-MS)and multistage mass spectrometry (MSn), we identified the glycosylation sites inβ-conglycinin. According to the ESI-MS results,the structure ofN-glycans and the molecular weight of glycopeptides were identified. Data analysis strategy is shown in Fig. 1.For example, according to the 202 Da molecular weight difference between glycopeptide treated by TFMS and PNGase F, NLKT and KVANKT were identified to be glycopeptides which modified by glycans (H5N2 and H5N4A2). But the specific combination between peptides and glycans has remained unclear. There were four possible glycopeptide combinations, as NLKT-H5N2, NLKTH5N4A2, KVANKT-H5N2, and KVANKT-H5N4A2, which leads to four kinds of molecular weight: 1 691 [M+H]+, 1 431 [M-2H]2-,1 338 [M-2H]2-, and 1 876 [M+H]+. According to the ESI-MS profiles of glycopeptides, there were only two peaks (1 691 [M+H]+,1 431 [M-2H]2-) appeared, illustrating the exact combination of glycans and peptides could be clarified. To be specific, NLKT was modified by H5N2, and KVANKT was modified by H5N4A2.
To confirm the accuracy of the analytical strategy we used,N-glycosylation sites on the standard sialylated glycopeptide and their corresponding glycan structures were analyzed by the strategy mentioned above. The results can be found in the supporting Fig. S1.Furthermore, the structure of glycopeptides existed inβ-conglycinin were identified by ESI-MS/MS (data were presented in supporting file Figs. S2-S7), and theN-glycosylation sites and their corresponding glycan structures were specifically analyzed.

2.3 Purification of soybean β-conglycinin
Soybeans were grounded with liquid nitrogen and passed through a 60-mesh sieve, then acetone and ethyl ether were used to remove fat alternately for four times. Defatted soybean powder was dissolved in Tris-HCl (30 mmol/L, pH 8.0) at a ratio of 1:15 (m/V), and stirred for 1 h at room temperature. The total protein in soybean was dissolved in the Tris-HCl.
Soybean 7S globulin was separated from total soy protein by isoelectric precipitation following the method of Picariello et al. [22]with little modifications. The mixture of total protein was centrifuged for 30 min (9 000 ×g), and the pH of the supernatant was adjusted to 6.4. Then the mixture was stored at 4 °C overnight and then centrifuged (9 000 ×g) for 20 min. After removed the sediment, NaCl powder was added to the supernatant and the final concentration of NaCl was 0.25 mol/L, then HCl (2 mol/L) was used to adjust the pH to 5.5. The mixture was stirred at 4 °C for 30 min and then centrifuged(9 000 ×g, 4 °C) for 30 min to remove the sediment. After that, the supernatant was diluted twice by double distilled water, and then the pH of the diluent was adjusted to 4.8. The mixture was stirred at 4 °C for 30 min and then centrifuged (9 000 ×g, 4 °C) for 30 min to remove the sediment. At this time, the supernatant was neutralized by NaOH (2 mol/L), then lyophilized and stored at -20 °C. The protein powder is soybean 7S globulin.
Soybean 7S globulin was dissolved in phosphate buffer(2.6 mmol/L NaH2PO4, 32.5 mmol/L Na2HPO4, 0.4 mol/L NaCl, pH 7.6)at a ratio of 1:5 (m/V), and then loaded to a Sepharose CL-6B column(2.5 cm × 1.2 m). The flow rate of elute buffer is 0.15 mL/min,β-conglycinin was eluted by the same phosphate buffer and collected in the receptor machine. The content of total soy protein, soybean 7S globulin andβ-conglycinin was tested by BCA protein assay kit.
2.4 Sodium dodecyl sulfonate polyacrylamide gel electrophoresis (SDS-PAGE)
Samples were prepared at a concentration of 1 mg/mL and mixed with the loading buffer and boiled at 100 °C for 5 min. The experiment was performed in a mini-protean II gel apparatus (Bio-Rad,USA), by using a separating gel of 12% acrylamide and a stacking gel of 4% acrylamide for separation. The gel was stained with Coomassie Brilliant Blue R-250 for 40 min, and bleached in destaining solution(1.31 mol/L HAc, 1.23 mol/L methanol) for 12 h. Bio-Rad GelDoc 2000 gel imaging system was used to photograph and analysis the electrophoresis results [23].
2.5 Enzymatic hydrolysis of soybean β-conglycinin by trypsin and pepsin
Five milligrams of soybeanβ-conglycinin globulin were added to 0.2 mL 10 mmol/L dithiothreitol (DTT) and reacted at 60 °C for 40 min.Then 1.84 mg indole-3-acetic acid (IAA) was added and the mixture was incubated in dark for 1 h. After that, 6-fold volume of acetonitrile was added and the mixture was stored at 4 °C for 2 h and then centrifuged at 12 000 ×gfor 3 min. The supernatant was removed and the protein was freeze-dried. Then 1 mL ammonium bicarbonate buffer (50 mmol/L,pH 7.5) and 0.1 mg trypsin enzyme (10 000 BAEE units/mg) were mixed with the protein powder and the mixture was incubated overnight at 37 °C [24]. The reaction was then stopped by heating to 95 °C for 10 min and the mixture was freeze-dried and re-dissolved in 1 mL hydrochloric acid buffer solution (pH 2).Then 5 mg pepsin (500 U/mg) was added and the mixture was stored overnight at 37 °C followed by heating at 100 °C for 5 min. The pH was adjusted to neutrality by 2 mol/L NaOH and the mixture was centrifuged at 12 000 ×gfor 3 min. The supernatant was then freezedried to obtain the peptide fraction as described by Keshavarz et al. [25].
2.6 Glycopeptide enrichment and purification
One hundred milligrams medical-grade absorbent cotton was evenly packed into the 1 mL pipette tip and the cotton column was cleaned with 10 mL H2O and 100% acetonitrile. Peptides digested by trypsin and pepsin were dissolved in 1 mL H2O and centrifuged at 12 000 ×gfor 3 min. The supernatant was added to the cotton column. Impurities on the cotton column were eluted orderly by 15 mL of 100% (V/V), 95% (V/V),85% (V/V), and 75% (V/V) aqueous acetonitrile, respectively. Six milliliters of H2O was used to elute the glycopeptide and eluates were freeze-dried at -20 °C for subsequent ESI-MS and MS/MS analyses.
2.7 Removal of N-glycans by TFMS
As Chen et al. [26] described earlier, 5 mg of the purified glycopeptide were placed in a 4 mL reaction flask to which 1 mL TFMS containing 5% (V/V) toluene was added. The reaction bottle was then filled with nitrogen at -20 °C for 100 min.Pyridine:methanol:water (3:1:1,V/V) mixture was then added to quench the reaction, and 0.5% ammonium bicarbonate was added to adjust the pH to 7.0. The solution was concentrated, dialyzed, and lyophilized for subsequent ESI-MS and MS/MS analysis.
2.8 Release of N-glycans by PNGase F
According to the methods reported by Hua et al. [27], 5 mg of glycopeptide samples were dissolved in 50 µL H2O to which 50 µL denaturation solution (0.17 mol/L SDS and 0.4 mol/L DTT) was added. The mixture was then heated at 100 °C for 10 min. Samples was then cooled to 25 °C, and 60 µL enzymolysis buffer, which composed of 0.5 mol/L sodium phosphate solution (pH 7.5), 60 µL NP-40 (nonionic surfactant), and 1.5 µL PNGase F (500 U/µL), was added to the system. The reaction was proceeded at 37 °C for 24 h.After centrifuged at 12 000 ×gfor 3 min,N-glycans were purified by C18and porous graphitized carbon (PGC) extraction columns for subsequent ESI-MS analysis.
2.9 ESI-MS and MS/MS analysis
According to Li et al. [28], the ESI-MS parameters were set as follows: the quantitative loop of the mass injection inlet was 2 µL,methanol:water (1:1,V/V) mixture was used as the mobile phase and the flow rate was set as 200 µL/min, the capillary temperature was 300 °C, and the injection rate was 20 µL/min, the working voltage of ion source electrospray was 4 kV, the capillary lens voltage was 250 V,the ratio of the auxiliary gas flow rate to the sheath gas velocity was 1:2, the auxiliary airflow speed was 3 L/min, the capillary voltage was 37 V, and the data were acquired with LTQ Tune (Thermo Fisher Scientific, Waltham, MA, USA). The MSndetection parameters were set as follows: the colliding gas was He, the slit width was 3 Da, and the collision energy was set as 30 eV.
2.10 Database retrieval and peptide ion fragment analysis
The MS/MS profiles of peptides were searched in Mascot database (Matrix Science, Boston, MA, USA). Experimental parameters were set as follows: the fragment ion mass error was± 0.5 Da, the peptide parent ion tolerance was 0.7 Da, the protease was selected as trypsin, the fixed modification was cysteine iodoacetamide(+ 57 Da), the variable modifications were methionine oxidation(+ 15.9 Da), GlcNAc (+ 203 Da), and the deamidation group(+ 0.98 Da), and the fragment ion was the b and y series. Besides, the amino acid sequence of glycopeptides was compared with the linear epitopes profile ofβ-conglycinin [29,30].
2.11 Spatial structure analysis
Spatial structure ofβ-conglycinin was built on the PyMOL software (http://www.pymol.org/) usingβ-conglycinin (PDB database, http://www.rcsb.org/structure/1IPK) as a model [31]. The lineal epitopes and glycosylation sites located in allergic epitopes were marked in blue and red separately.
3. Results and discussion
3.1 Analysis of the glycosylation sites and corresponding glycan structure of soybean β-conglycinin
We used 10 g of soybean to extract the allergenβ-conglycinin,and the content of total soybean protein, 7S globulin andβ-conglycinin was defined by BCA protein assay kit. Bovine serum albumin was used as standard and the standard curve was presented in Fig. 2A.According to Fig. 2B, there were 3.18, 1.04, 0.76 g of total protein, 7S globulin andβ-conglycinin existed in the soybean, and the extraction rate ofβ-conglycinin is 7.6%. Three peaks were appeared in the chromatography, andβ-conglycinin were identified in both peak 1 and peak 2 (Fig. 2C). There wereα’,α, andβsubunits in the SDS-PAGE pattern (peak 1 and peak 2). Low molecular weight proteins were eluted in peak 3 (Fig. 2D).
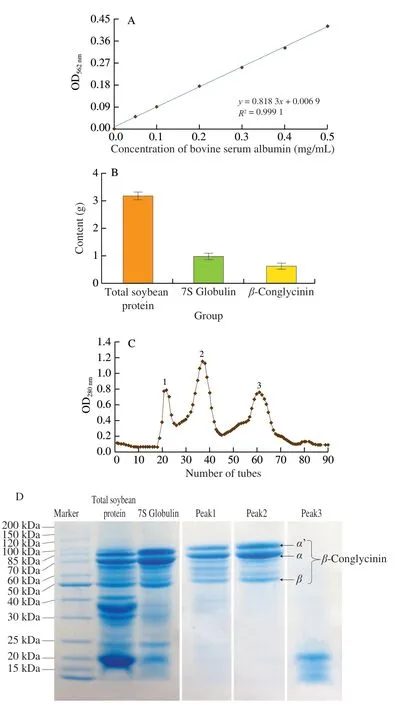
Fig. 2 The content of soybean allergen β-conglycinin in 10 g of soybean.(A) The standard curve of BCA assay kit. (B) Content of total soybean protein,7S globulin and β-conglycinin. (C) Elute curve of agarose gel chromatography.(D) SDS-PAGE of total soybean protein, 7S globulin and β-conglycinin.
β-Conglycinin from total soybean protein were purified and then digested by trypsin and pepsin. Glycopeptides were enriched on a cotton hydrophilic column, and analyzed by ESI-MS and MS/MS. As shown in Fig. 3, there were 22 peaks appeared, indicating that there were 22 kinds of glycopeptides existed in the system. There are three questions need to be further explored. Firstly, the glycosylation sites need to be localized. Secondly, the structure ofN-glycans attached to the glycopeptides need to be analyzed. And the specific combinations between peptides and glycans need to be identified.
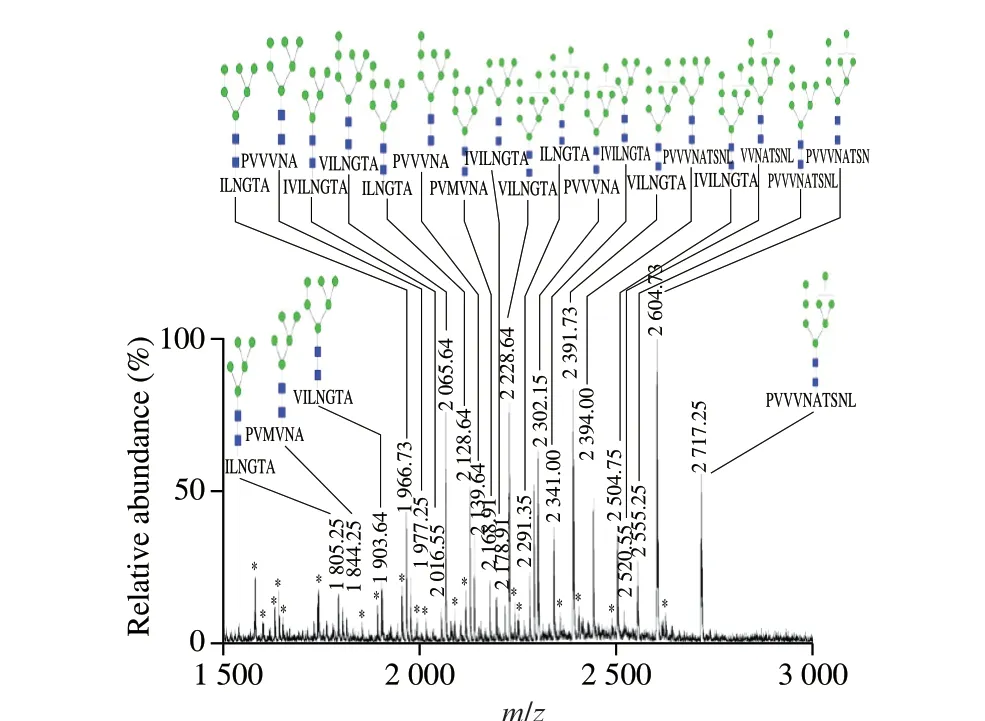
Fig. 3 ESI-MS spectra of soybean β-conglycinin hydrolyzed by trypsin and pepsin. Blue squares, N-acetyl-D-glucosamine; green circles, mannose;*, non-glycopeptide peaks.
For learning the comprehensive information in glycopeptides,glycans need to be released from the glycopeptides at first. Enzymatic and chemical methods were commonly used in this process. For example,N-glycans could be released from glycopeptides by PNGase F unless the GlcNAc in the pentose core was modified byα-1,3-fucose [32].During the release process, PNGase F could transform the asparagine to aspartic acid, leading to a 0.98 Da increase of molecular weight [33]. And a 2.98 Da increase in molecular weight could be induced if the reaction is conducted in the presence of H218O [34].Endo-β-N-acetylglucosaminidase-H (Endo H) could break the glucosidic linkages between GlcNAc in the pentose core and generate a quality label (+ 203 Da) at the glycosylation site. However, it could only act on heterozygousN-glycans andN-glycans which is rich in mannose [35]. Comparably, the TFMS method is universal and highly efficient [36]. It could release all types of glycans and has been used in glycoproteomics researches [36]. We have noticed that C-Nlinked amide bond is stable while the C-O-linked glycoside bond is easy to break under acidic conditions, elucidating that interference fromO-glycans may be avoided when glycopeptides are released by TFMS. A GlcNAc (+ 203 Da) at theN-glycosylation site could also be formed and set as a quality label, which subsequently reinforced the accuracy of the localization of glycosylation sites by using mass spectrometry [37]. By calculating the molecular weight of glycans and glycopeptides, the actual combination between peptides and glycans could be defined.
TFMS and PNGase F were then used to hydrolyze the glycopeptide separately in this research. As shown in Fig. 4,there were nine peptides detected in both spectrums, indicating that both TFMS and PNGase F could releaseN-glycans from glycopeptides effectively. Compared to peptides hydrolyzed by PNGase F (Fig. 4B), a 202 Da increase in the molecular weight were appeared when glycopeptides were treated by TFMS (Fig. 4A), for the GlcNAc was conserved as the quality label. To know the amino acid sequence of glycopeptides, MS2were then performed. In the following experiment, we takem/z1 238.42 and 813.25 for example,and describe the identification process of glycosylation site in detail.The sequence of amino acids in other glycopeptides were analyzed in the same way.
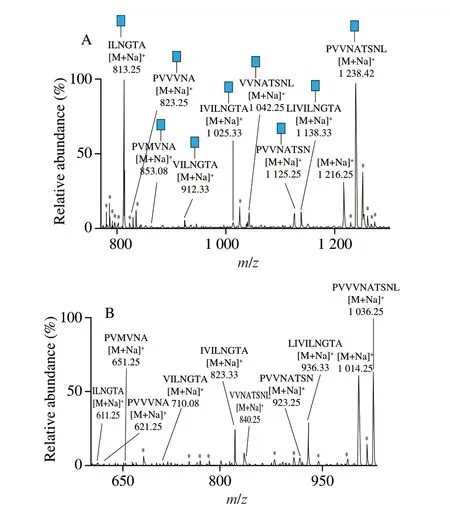
Fig. 4 ESI-MS spectra of deglycosylated soybean β-conglycinin.(A) Glycopeptide deglycosylated by TFMS. (B) Glycopeptide deglycosylated by PNGase F. Blue squares, N-acetyl-D-glucosamine; *, non-glycopeptide peaks.
To localize the glycosylation site, TFMS were used to break the glycosidic bonds, and glycopeptides which contain a GlcNAc was formed and then analyzed by MS/MS. As shown in Fig. 5A,m/z1 011.33 (b8) represents for the fragment which derived from them/z1 125.33 (b9) with an asparagine residue (114 Da) removed,illustrating the amino acid located in b9is asparagine. Similarly, the entire amino acid sequence of glycopeptides could be defined by comparing the molecular weight of different peaks. To identify the glycosylation sites, we compared the molecular weight of b5-H2O(m/z734.25) and b4(m/z435.08), clearly there is 299 Da decrease caused by a GlcNAc residue (203 Da) and asparagine residue(114 Da). This result illustrated that the glycan was attached to the asparagine residue, which located in the b5position. By using this method, the amino acid sequence of glycopeptide (m/z1 238.42)is deducted to be PVVVN#ATSNL, where#represents for the glycosylation site.
The amino acid sequence of glycopeptide (m/z813.25) was analyzed in the same way. As shown in Fig. 5B,m/z587.25 (y4) was calculated to bem/z252.00 (y3-H2O) plus the molecular weight of asparagine (114 Da), H2O (18 Da) and GlcNAc residue (203 Da).This result illustrated that the glycan was attached to the asparagine residue, which located in the y4position. The amino acid sequence was deducted in the same manner, and the amino sequence ofm/z813.25 was identified as ILN#GTA, where#represents for the glycosylation site. In this way, all the amino sequence of glycopeptides were identified, and the MS/MS spectrums for other glycopeptides could be found in supporting information (Figs. S2-S7).
We used PNGase F to release all the glycans from soybeanβ-conglycinin and identified glycan structures by ESI-MS. The results are shown in Fig. 5C. Four kinds of high-mannoseN-glycans were identified. According to the ESI-MS spectrum in Fig. 3, clearly that there are 22 kinds glycopeptides existed in theβ-conglycinin. Like we mentioned in the experimental strategy, there would be many possible combinations between peptides and glycans, and the total molecular weight of the specific combination could be calculated.The actual structure of glycopeptides could be identified only when the total molecular weight appears in the ESI-MS spectrum in Fig. 3.According to this principle, we calculated the molecular weight of all the possible combinations between glycans and peptides, compared the results with the peaks in Fig. 3, and identified the structure of 22 kinds of glycopeptides.
Furthermore, the amino acid sequence ofα(GenBank:QBZ93318.1),α’ (NCBI reference sequence: NP_001237316.2)andβ(NCBI reference sequence: NP_001236872.2) subunits in soybeanβ-conglycinin were acquired from National Center for Biotechnology Information (NCBI) website (http://www.ncbi.nlm.nih.gov/). And in this study, both the amino acid sequence of glycopeptides and glycosylation site were identified by MS/MS.Then, the obtained amino acid sequence of glycopeptides was searched in the protein database using Mascot software, and these glycosylation sites inβ-conglycinin subunit position were determined.By comparing the amino acid sequence inα,α’,βsubunits and the glycosylation sites, 5 glycosylation sites were identified.
To be specific, glycopeptides inαsubunit, which include ILNGTA, VILNGTA and IVILNGTA, are glycosylated in Asn-199 and modified by H5N2, H6N2, H7N2 and H8N2. Other glycopeptides inαsubunit ofβ-conglycinin, which include PVVVNA and PVVVNATSNL, are glycosylated in Asn-455 and modified by H6N2,H7N2 and H8N2. Glycopeptides inα’ subunit ofβ-conglycinin,which include ILNGTA, VILNGTA and IVILNGTA, are glycosylated in Asn-215 and modified by H5N2, H6N2, H7N2 and H8N2. Another glycopeptide PVMVNA located in theα’ subunit ofβ-conglycinin was glycosylated in Asn-489, and modified by H5N2 and H7N2. Glycopeptide VVNATSNL is located in theβsubunit ofβ-conglycinin, glycosylated in Asn-326, and modified by H8N2. We compared our results with the reported researches on the glycosylationinformation of soybean allergenβ-conglycinin [38], and we are sure that our results are consistent with these researches.N-glycosylation sites and the structures of corresponding glycans inβ-conglycinin were listed in Table 1.

Table 1 N-Glycosylation sites and corresponding glycan structures of soybean β-conglycinin.

Fig. 5 MS/MS spectrums of soybean β-conglycinin. (A) m/z 1 238.42 (PVVVN#ATSNL). (B) m/z 813.25 (ILN#GTA). (C) ESI-MS spectra of the β-conglycinin N-glycans. Blue squares, N-acetyl-D-glucosamine; green circles, mannose; *, non-glycopeptide peaks and non-glycan peaks.
3.2 Analysis of the glycosylation site and corresponding glycan structure of β-conglycinin
The spatial structure ofβ-conglycinin was modeled and presented in Fig. 6, 10 linear epitopes were marked in blue, and glycosylation site located in the epitopes were marked in red. As seen, there is one glycopeptide appeared in the liner epitopes started from Ala-344 and ended in Leu-359 inβ-conglycinin, the amino acid sequence of the glycopeptide is VVN#ATSNL, where#represents for the glycosylation site. It can be assumed that high-mannose oligosaccharides, which connected to the linear epitope VVNATSNL, would prompt the antigenpresenting process proceeded by dendritic cells.
To be specific, it has been reported that glycopeptides modified by high-mannose glycans would maintain enhanced targeting to the human APCs mediated via the mannose receptor, and APCs could deliver the immune-signals in a more efficient way [39]. According to McIntosh’s research [40] on human cytomegalovirus (CMV) tegument glycopeptides pp65, the binding between glycopeptides and APCs increased significantly when compared with the non-glycosylated control. And glycopeptides modified by high-mannoseN-glycans at a single site outside the T cell epitope were presented by the APCs and activated the T cell clone. Compared with non-glycosylated peptides,glycopeptides modified by high mannoseN-glycans could also induce the maturation of dendritic cells, which is usually act as APCs, and further prompt the binding between glycopeptides and APCs [41].However, there is another research find that N-linked glycosylation increased efficacy of malaria vaccine by improving immunogenicity and/or focusing the response towards relevant epitopes by glycan masking [42]. According to Boes’s research [42],a number of epitopes were shielded from immune recognition by high-mannosetypeN-glycans, illustrating the possibility that glycosylation can be used for the directed targeting of immune responses.N-glycans attached to the allergic glycopeptides are important for the development of food allergy, and their specific functions on the antigen presentation and degranulation need to be further explored.
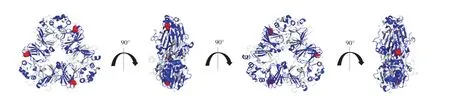
Fig. 6 The ribbon presentation of linear epitopes and glycosylation sites in β-conglycinin. The basic structure of peptides was marked in gray, epitopes were marked in blue, glycosylation sites located in the epitopes was marked in red.
According to our results presented in Table 1, information about glycosylation sites andN-glycans on theβ-conglycinin were clarified.We are planning to remove all theN-glycans fromβ-conglycinin,and study the variation in antigen-presenting process and antibodybinding process in the following experiment. Based on our results,vaccines and/or therapeutic agents targeting on the glycans which bond to theβ-conglycinin, could be developed. Besides,the glycosylation information could also help to proceed the mechanism study focused on the relationship between allergens’structure and immunoreactivity.
4. Conclusions
In the present study, we utilized cotton hydrophilic chromatography to enrich glycopeptides, and TFMS was utilized to release glycans from glycopeptides.N-glycosylation sites and their correspondingN-glycan structures ofβ-conglycinin were identified by using ESI-MS and MS/MS. As a result, five glycosylation sites were identified. Two glycosylation sites located inαsubunit ofβ-conglycinin, Asn-199 and Asn-455, were identified.And 2 glycosylation sites located inα’ subunit ofβ-conglycinin,Asn-215 and Asn-489, were localized. Inβsubunit ofβ-conglycinin,Asn-326 were identified. All the identified glycosylation sites were attached to high-mannoseN-glycans. It is worth noticing that glycopeptides VVN#ATSNL (where#represent for the glycosylation sites), which modified by H8N2 and located inβsubunit ofβ-conglycinin, is also a linear epitope. High-mannoseN-glycans which attached to the epitope were assumed to prompt the antigenpresenting process. Our study provided a methodological reference for the identification and analysis of glycosylation information of various food allergens, the clarification of glycosylation structures of food allergens would help to understand the molecular mechanism of food allergy.
Conflict of interest
The authors declare that this manuscript has no conflict of interest.
Acknowledgements
This research was funded by National Natural Science Foundation of China (31870798, 31972024), Shaanxi Province Innovation Capability Support Plan-Science and Technology Innovation Team (2020TD-044), Key Laboratory of Glycobiology and Glycoengineering of Xi’an (2019219514SYS010CG032), Natural Science Project of Shaanxi Provincial Department of Education(21JK0929), and Science and Technology Resources Sharing Platform Project of Science and Technology Department in Shaanxi Province(2022PT-46).
Appendix A. Supplementary data
Supplementary data associated with this article can be found, in the online version, at http://doi.org/10.1016/j.fshw.2022.09.025.
杂志排行
食品科学与人类健康(英文)的其它文章
- The role of probiotics in prevention and treatment of food allergy
- Roles of fermented plant-, dairy- and meat-based foods in the modulation of allergic responses
- The role of gut microbiota and its metabolites short-chain fatty acids in food allergy
- Association of nutrients intake during pregnancy with the risk of allergic disease in offspring: a meta-analysis of prospective cohort studies
- Purif ication and immunoglobulin E epitopes identif ication of low molecular weight glutenin: an allergen in Chinese wheat
- Determination of egg and milk allergen in food products by liquid chromatography-tandem mass spectrometry based on signature peptides and isotope-labeled internal standard